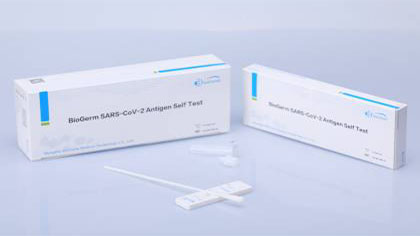
BioGerm News丨BioGerm 2019-nCoV Antigen Self Test Kit (Colloidal Gold) Granted EU CE Certification!
Recently, an outstanding newcomer was incorporated into BioGerm CE certificated family - BioGerm 2019-nCoV Antigen Self Test Kit (Colloidal Gold) was granted EU CE certification, showing that the quality of BioGerm medical products is recognized and ratified by international authorities once again.
A newcomer of overseas products
Recently, BioGerm 2019-nCoV Antigen Self Test Kit (Colloidal Gold) was granted EU CE certification, indicating an entry criteria compliance for EU market. In the EU market, the “CE” mark is a compulsory certification mark for a product to circulate freely in the EU market to show that the product meets the compliance requirements of the relevant EU directives on medical devices.
The CE certification shows that the quality of BioGerm medical products is recognized and ratified by international authorities once again, which is a leverage for BioGerm to enhance the international competitiveness and facilitate the prevention and control of COVID-19 worldwide.

BioGerm 2019-nCoV Antigen Self Test Kit (Colloidal Gold) was Granted EU CE Certification
Outstanding performance of BioGerm 2019-nCoV Antigen Test Kit
The 2019-nCoV Antigen Self Test Kit (Colloidal Gold), developed independently by BioGerm, features high sensitivity and precision. Missed test is reduced in the kit substantially. It is able to detect infected patients with low 2019-nCoV viral load at the early stage to curb the spread of the virus effectively. In terms of test time, test results can be obtained in 10-20 min;

In the CoA issued by BIOMEX medical lab, the BioGerm 2019-nCoV Antigen Test Kit, with a sensitivity of 97.17% and a specificity of 99.68%, is highly appraised and recommended.

According to the laboratory study, the BioGerm 2019-nCoV Antigen Self Test Kit (Colloidal Gold) is able to detect common variants, including Delta and Omicron, effectively.
Product Characteristics:
Rapid test speed: Test results can be obtained in 10-20 min;
Flexible samples to be detected: Human oropharyngeal swabs, nasopharyngeal swabs, and saliva samples are suitable;
Handy operation: Test results can be obtained through visual interpretation directly;
Easy storage: It can be stored and shipped at 2 ℃ -30℃.
BioGerm has been devoting to providing a comprehensive product line for global anti-pandemic action. The CE certified Nucleic Acid Test Integral Solution Product Line covers a whole process of 2019-nCoV test products required for sampling, extraction, amplification and other procedures; the successive approval of products of colloidal gold line has perfected BioGerm 2019-nCoV Whole Setting Solution. The detection window is shortened and the positive detection rate is improved through the combined application of multiple test methods concerning nucleic acid, antigen and antibody, to achieve an accurate and rapid test and facilitate the prevention and control of the global epidemic.